
License: CC BY-SA: Attribution-ShareAlike
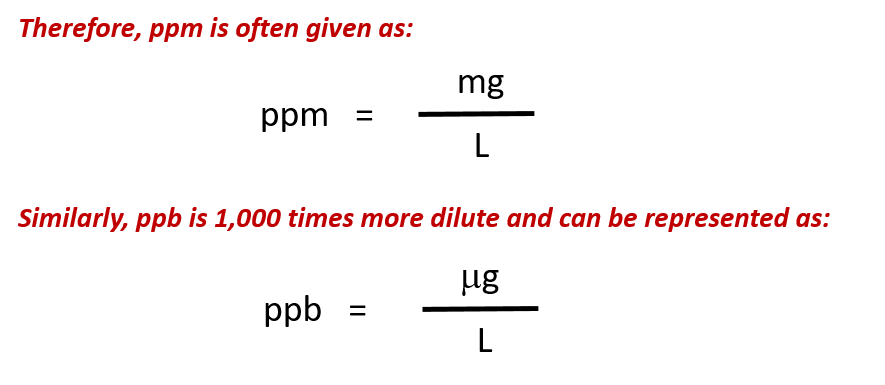
Dilution-concentration simple example.License: Public Domain: No Known Copyright. Solving Dilution Problems in Solution Chemistry CLEAR & SIMPLE - YouTube.To sum up: we converted to each reactant’s moles by using the given concentrations as conversion factors, expressing molarity as mol/L once we found our limiting reactant, we converted through to grams of AgCl formed.ĬC licensed content, Specific attribution Therefore, 17.6 g AgCl( s) is formed in the reaction. The volume of solvent needed to prepare the desired concentration of a new, diluted solution can be calculated mathematically. For the solutions to be usable in the lab (for a titration, for instance), they must be accurately diluted to a known, lesser concentration. Diluting solutions is a necessary process in the laboratory, as stock solutions are often purchased and stored in very concentrated forms. Dilution can also be achieved by mixing a solution of higher concentration with an identical solution of lesser concentration. This process keeps the amount of solute constant, but increases the total amount of solution, thereby decreasing its final concentration.
How to calculate ppm of iron using tha mass value serial#
serial dilution: stepwise dilution of a substance in solutionĭilution refers to the process of adding additional solvent to a solution to decrease its concentration.dilution: a solution that has had additional solvent, such as water, added to make it less concentrated.A serial dilution is a series of stepwise dilutions, where the dilution factor is held constant at each step.Dilution calculations can be performed using the formula M 1V 1 = M 2V 2.When calculating dilution factors, it is important that the units of volume and concentration remain consistent. Most commonly, a solution ‘s concentration is expressed in terms of mass percent, mole fraction, molarity, molality, and normality.

